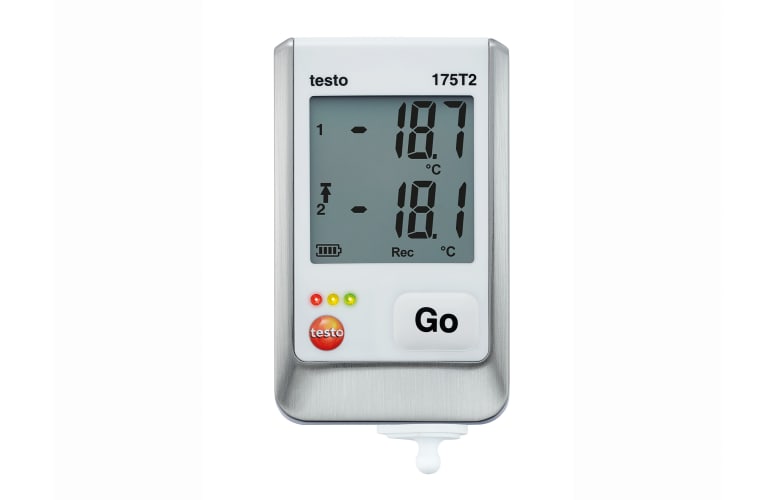
-
Intuitive and validatable software for the pharmaceutical industry
-
Graphic and tabular display of all measurement parameters
-
Effortless creation and management of reports
-
Digital signature and audit trail for maximum security and safety
The ComSoft 21 CFR Part 11 software means you get validatable software for programming and readout of Testo data loggers and for archiving data. It fulfils all the FDA's 21 CFR Part 11 requirements.